Electrochlorination technology has emerged as a critical solution for global water disinfection challenges, particularly as freshwater scarcity intensifies worldwide. This electrochemical process transforms simple salt solutions into powerful disinfectants, offering a sustainable alternative to traditional water treatment methods. With applications spanning from municipal drinking water to industrial wastewater treatment, electrolytic chlorine generation represents a convergence of electrochemistry, materials science, and environmental engineering. The technology continues to evolve rapidly, driven by material innovations, renewable energy integration, and the urgent need for cost-effective water disinfection solutions. As we examine its principles, advantages, applications, and future trajectories, we gain insight into why electrochlorination has become an indispensable technology for securing water safety in an increasingly water-stressed world.
1 Basic Principles of Electrochlorination
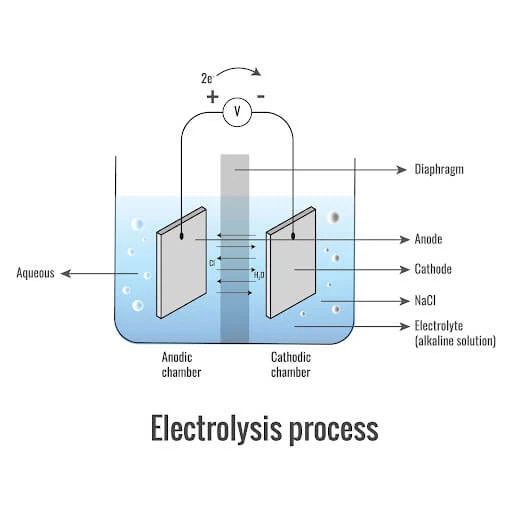
Electrochlorination operates on fundamental electrochemical principles where electrical energy drives chemical transformations. The core process involves passing direct current through a sodium chloride (NaCl) solution, initiating redox reactions at the electrodes. At the anode, chloride ions undergo oxidation: 2Cl⁻ → Cl₂ + 2e⁻, generating chlorine gas. Simultaneously at the cathode, water molecules are reduced: 2H₂O + 2e⁻ → H₂ + 2OH⁻, producing hydrogen gas and hydroxide ions 【1】【5】. The overall reaction can be summarized as:
2NaCl + 2H₂O → Cl₂ + H₂ + 2NaOH
The chlorine gas produced immediately hydrates in water to form hypochlorous acid (HOCl), the primary disinfectant responsible for microbial inactivation. This equilibrium depends on pH:
Cl₂ + H₂O ⇌ HOCl + H⁺ + Cl⁻
HOCl ⇌ H⁺ + OCl⁻
The proportion of HOCl (more effective disinfectant) to OCl⁻ decreases as pH rises above 7.5. In seawater electrolysis, which contains approximately 19g/L chloride ions, the process yields a sodium hypochlorite solution directly through the reaction:
NaCl + H₂O → NaClO + H₂ 【10】
System efficiency depends critically on several parameters:
Electrolyte concentration (typically 2.5-4.0% brine solution)
Current density (optimized to balance reaction rate and electrode longevity)
Temperature (affects reaction kinetics and solubility of byproducts)
Electrode material (determines overpotential and catalytic activity)
Modern systems employ specialized ion-exchange membranes that selectively permit cation transport while preventing chlorine-hydrogen mixing, significantly enhancing safety and purity 【1】. This fundamental electrochemical framework enables the sustainable production of disinfectants using only salt, water, and electricity as inputs.

2 Technical Advantages of Electrochlorination
Electrochlorination systems offer compelling advantages over conventional chlorine-based disinfection methods, driving their widespread adoption across water treatment applications:
Enhanced Safety Profile: Traditional chlorination requires transporting and storing large quantities of hazardous liquefied chlorine gas. Electrochlorination eliminates these risks by generating chlorine on-site and on-demand, significantly reducing hazards associated with transportation, storage, and handling of chlorine gas. This "just-in-time" production model minimizes community exposure risks and regulatory compliance burdens 【1】【5】.
Operational Simplicity and Cost Efficiency: The primary feedstock-sodium chloride-is inexpensive and globally available. Coastal facilities can directly utilize seawater, virtually eliminating raw material costs. Modern electrochlorination systems feature automated control systems that continuously monitor and adjust chlorine production based on water quality parameters, reducing operational labor requirements. System designs have evolved toward modular "plug-and-play" configurations that simplify installation and scalability 【5】【9】.
High Disinfectant Purity and Efficacy: Electrochemically generated chlorine contains fewer impurities compared to commercially supplied liquefied chlorine or bleach products. The resulting disinfectant solution maintains consistent high efficacy against diverse pathogens including bacteria, viruses, and protozoa. This purity advantage translates to predictable disinfection performance and minimizes unwanted side reactions that form problematic disinfection byproducts (DBPs) like trihalomethanes 【1】.
Environmental Compatibility: Electrochlorination avoids the carbon footprint associated with manufacturing and transporting traditional chlorine products. When powered by renewable energy sources, the technology offers a carbon-neutral disinfection alternative. Furthermore, the process co-produces hydrogen gas at the cathode, which can potentially be captured and utilized as clean fuel, enhancing overall system sustainability 【5】【9】.
3 Major Application Fields
Electrochlorination has established critical roles across diverse water treatment sectors, each leveraging its unique advantages:

Municipal water treatment represents the most significant application of electrochlorination technology. Large-scale systems provide primary or residual disinfection for urban water supplies, effectively controlling pathogens throughout distribution networks. This installation demonstrates the technology's scalability, reliability, and cost-effectiveness for major urban water infrastructure 【1】. Smaller-scale systems are increasingly deployed in rural communities and developing regions where bulk chlorine delivery is impractical or uneconomical.

Electrochemical chlorine generation effectively addresses complex wastewater challenges through two primary mechanisms: disinfection and advanced oxidation. Beyond pathogen destruction, electrochlorination systems generate powerful oxidants that degrade organic pollutants and eliminate color compounds in textile effluents. The technology demonstrates particular effectiveness in treating recalcitrant contaminants in industrial wastewater streams, significantly enhancing biodegradability before biological treatment stages. This dual-action capability-disinfection plus chemical oxidation-provides comprehensive treatment while minimizing chemical storage requirements 【1】.

Industrial cooling systems, particularly in power generation and petrochemical facilities, face persistent biofouling challenges from microorganisms in cooling water. Biofilm formation on heat exchange surfaces dramatically reduces thermal efficiency and can promote corrosion. Electrochlorination systems provide continuous low-level chlorination (typically 0.2-1.0 ppm residual) that effectively controls bacterial, algal, and mollusk colonization without requiring hazardous chemical storage. Modern systems are integrated with real-time monitoring of oxidant demand, enabling precise dosage control that maintains efficacy while minimizing chemical usage and discharge impacts 【1】【10】.

The International Maritime Organization's Ballast Water Management Convention has driven significant adoption of electrochlorination technology in marine applications. Shipboard systems treat ballast water during loading operations to prevent translocation of invasive species. Electrochlorination's ability to utilize seawater directly makes it particularly suitable for this application. Systems are designed with multiple safeguards including neutralization and residual oxidant removal before controlled discharge at receiving ports 【10】.
| Application Sector | Primary Function | Typical Installations | Technology Features |
|---|---|---|---|
| Municipal Drinking Water | Primary/residual disinfection | Central water treatment plants | Large-scale (up to 100 kg Cl₂/hr), automated residual control |
| Wastewater Reuse | Pathogen destruction, trace contaminant oxidation | Municipal wastewater plants, industrial sites | Medium-scale (10-50 kg Cl₂/hr), oxidation optimization |
| Industrial Cooling | Biofouling control | Power plants, refineries, manufacturing | Continuous low-dose chlorination, seawater compatible |
| Ballast Water Treatment | Biocide treatment | Commercial vessels, naval ships | Compact systems, seawater electrolysis, discharge safeguards |
| Aquaculture | Disinfection in recirculating systems | Hatcheries, fish farms | Low-concentration applications, sensitive species protocols |
4 Research Progress and Technological Innovations
Recent advancements in electrochlorination technology focus on enhancing efficiency, durability, and environmental compatibility through materials science, process engineering, and system integration:

Traditional graphite electrodes have been largely replaced by dimensionally stable anodes (DSAs) based on titanium substrates coated with mixed metal oxides (RuO₂, IrO₂). These exhibit superior catalytic activity, corrosion resistance, and extended service life exceeding 5 years under continuous operation. Research demonstrates that nanostructured coatings with increased surface area can reduce chlorine evolution overpotential by 150-300 mV compared to conventional electrodes, significantly lowering energy consumption 【1】. Recent work with roughened silver-palladium cathodes (Pd/Ag(r)) demonstrates enhanced dechlorination capabilities for environmental remediation applications, achieving 85% yield in dechlorination of chlorinated organic compounds 【6】.
Direct seawater electrolysis faces persistent challenges from chloride-induced corrosion and competing reactions. The acidic-saline hybrid electrolysis system developed by Wang Jianlong and Guo Shaojun represents a significant breakthrough. This configuration couples acidic hydrogen evolution with saline chlorine evolution, reducing the cell voltage requirement to 1.59 V at 10 mA cm⁻²-a 27.7% reduction compared to conventional seawater electrolysis (2.20 V). Crucially, the acidified anolyte prevents formation of calcium and magnesium deposits that typically plague seawater systems. The approach demonstrates remarkable versatility, achieving high-efficiency E. coli and Staphylococcus aureus inactivation while enabling practical applications like pea sprout production using real-time generated disinfectant 【3】.
Traditional electrochlorination produces hydrogen as a byproduct, typically managed through dilution and venting. Innovative hybrid systems now enable simultaneous chlorine-free hydrogen production alongside wastewater treatment. The NiCo/MXene-based electrode system achieves hydrogen production at 9.2 mol h⁻¹ gcat⁻¹ with remarkably low electricity consumption (2.75 kWh per m³ H₂) at 500 mA cm⁻². This represents a 48% reduction in energy equivalent input compared to commercial alkaline water electrolysis. The integrated process simultaneously degrades hydrazine pollutants to ~3 ppb residual levels, demonstrating multi-pollutant treatment capability. These systems can be directly powered by low-voltage direct hydrazine fuel cells or solar cells, enabling self-sustaining operation 【4】.
The persistent challenge of chloride-induced electrode degradation in seawater applications has driven development of novel protective strategies. The NiFe layered double hydroxide electrocatalyst modified with poly(3,4-ethylenedioxythiophene):polystyrene sulfonate (NiFe LDH@PP/NF) creates an electrostatic repulsion barrier via sulfonate groups (-SO₃⁻) that effectively mitigates chloride intrusion. This innovation enables exceptional durability-continuous operation for 800 hours at 1000 mA cm⁻² and 300 hours at 2000 mA cm⁻²-with minimal performance degradation and negligible active chlorine production at the cathode. In situ Raman spectroscopy confirms that the modification promotes beneficial catalyst surface reconstruction while maintaining oxygen evolution efficiency 【8】.
5 Future Development Directions
As electrochlorination technology evolves, several strategic directions are emerging that will define its future trajectory:

Renewable Energy Integration: The inherent compatibility between electrochemistry and direct current from solar PV creates significant opportunities for off-grid and decarbonized water disinfection. Research focuses on designing systems with variable power input tolerance that can maintain stable chlorine output despite fluctuating renewable generation. Combined solar-electrochlorination installations are demonstrating viability for remote communities and humanitarian applications, eliminating dependence on chemical supply chains 【9】.
Advanced Electrode Development: Next-generation electrodes aim to achieve unprecedented efficiency and longevity through nanotechnology and materials engineering. Research priorities include:
Self-cleaning electrode surfaces that resist scale formation
Non-precious metal catalysts with performance comparable to RuO₂/IrO₂
Selective ion electrocatalysts that minimize oxygen evolution side-reaction
Bifunctional electrodes that alternate between chlorine production and electrochemical regeneration 【1】【8】
Smart System Architecture: The convergence of electrochlorination with digital technologies enables responsive, adaptive disinfection systems. Modern controllers incorporate machine learning algorithms that predict chlorine demand based on historical water quality parameters, flow rates, and seasonal variations. Sensor networks monitor multiple parameters including free chlorine, ORP, pH, conductivity, and DBPs precursors, enabling real-time process optimization. Cybersecurity considerations are increasingly integrated into control system designs for critical water infrastructure 【7】.
Application Expansion: Beyond traditional water treatment domains, electrochlorination shows promise in several emerging fields:
Precision agriculture: On-site generation of hypochlorous acid for irrigation system disinfection and pathogen control
Aquaculture: Water disinfection in recirculating aquaculture systems (RAS) with sensitive species
Hospital wastewater: Pathogen destruction in infectious waste streams
Hydroponics: Root zone disinfection without phytotoxicity concerns
Emergency response: Rapid deployment systems for disaster relief scenarios 【5】【9】
The trajectory of electrochlorination technology points toward increasingly efficient, intelligent, and sustainable water treatment solutions. As material innovations advance and renewable electricity costs continue to decline, electrochemical disinfection appears poised for expanded implementation across diverse sectors. However, ongoing research must address persistent challenges including electrode scaling in hard waters, management of disinfection byproducts under varying water quality conditions, and optimization for decentralized applications. The integration of electrochemical technologies with conventional water treatment represents a promising frontier for developing robust, multi-barrier treatment systems capable of addressing emerging contaminants and water scarcity challenges.
References
1.Electrolytic Chlorine Production Technology: Application and Development in Water Treatment. (2025). Baidu Academic. 1
2.Wang, J., & Guo, S. (2024). Energy-Efficient Electrosynthesis of High Value-Added Active Chlorine Coupled with H₂ Generation from Direct Seawater Electrolysis through Decoupling Electrolytes. Angewandte Chemie International Edition. 3
3.Yang, C., et al. (2025). PEDOT:PSS-Modified NiFe Layered Double Hydroxide Enables Efficient and Durable Seawater Electrolysis at High Current Density. Journal of Materials Chemistry A. 8
4.Energy-Saving Hydrogen Production by Chlorine-Free Hybrid Seawater Splitting Coupling Hydrazine Degradation. (2025). ProQuest. 4
5.Fundamental Principles and Applications of Electrochlorination. (2025). Jiangshitai. 5
6.Optimisation of Electrocatalytic Dechlorination of 2,4-Dichlorophenoxyacetic Acid on a Roughened Silver–Palladium Cathode. (2013). Electrochimica Acta. 6
7.Wuhan Xingda's New Patent Revolutionizes Electrolysis Systems. (2025). Sohu. 7
8.Technical Advantages and Future Development Prospects of Electrochlorination Technology. (2023). Hcbbs Forum. 9
9.Commissioning Technology for Seawater Electrochlorination Devices and Systems. (2023). 360Docs. 10
